Building
The plant was built on an area of 5417 square meters in distinguished area in the industrial city of Hassia according to the latest international standards, where the building was designed and equipped according to the requirements of the rules of good manufacturing Practices /cGMP/, in order to reach our scientific standard targets in production.
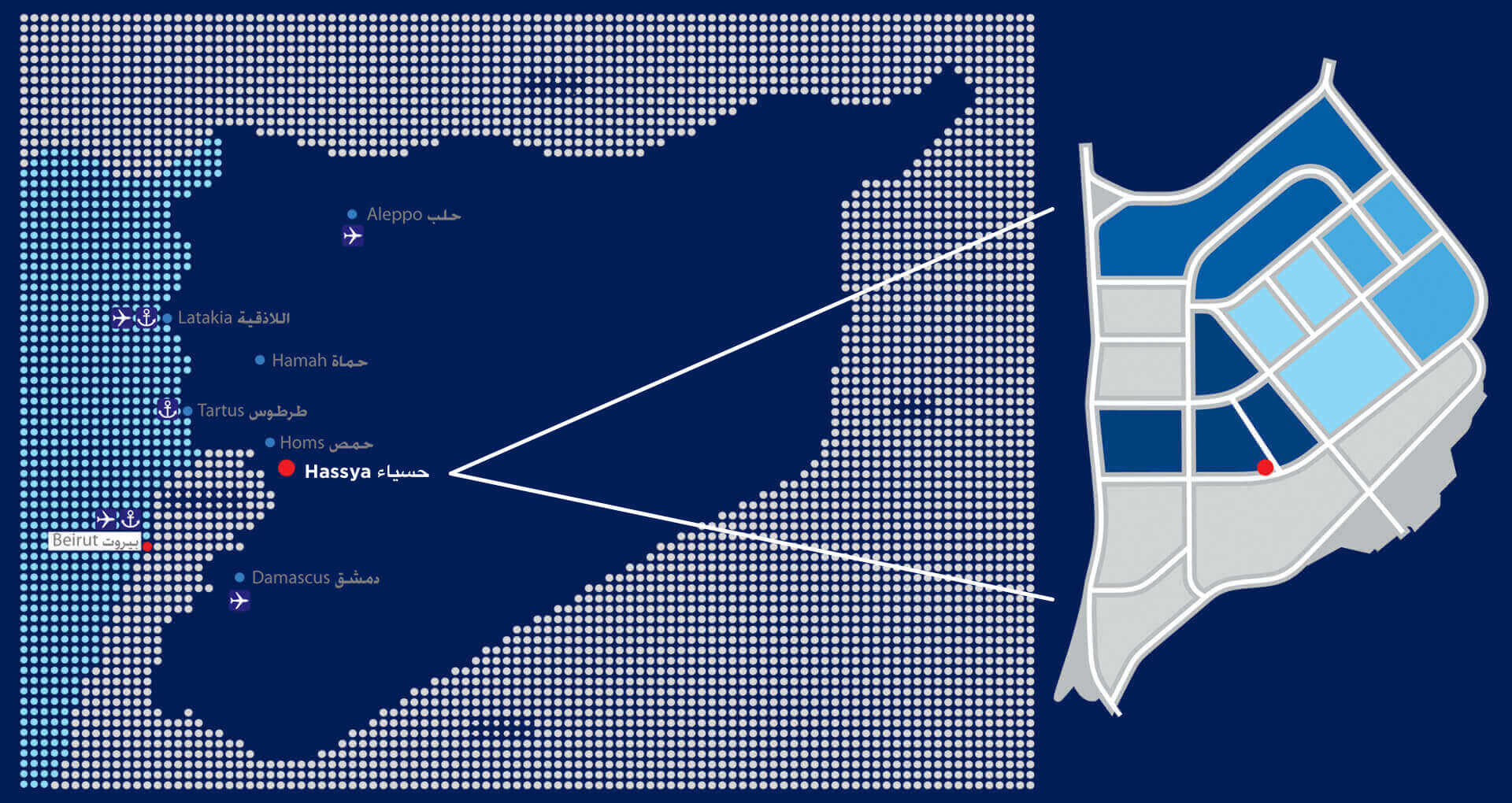
Location
After in-depth study carried out by the company, and found that the industrial city of Hassia is the best option as the location for the project Serda, because of its geographic expanding and its location close to markets and sea, air and land ports in Syria.
This city is distinguished by its infrastructure and interest from the Syrian government especially facilities provided like tax exemptions and benefits available for investment projects in this city.
And so was the choice of location in the area (food industries zone) within the city and close to the Free Zone Industrial Hassia.